Corrosion and coating options
Corrosion is a phenomenon that is universally known: iron and steel rust, copper greens, zinc bleaches, various forms of oxidation that no longer allow the metal to fulfil its role. Maintenance is then very costly and sometimes ineffective.
For the good preservation of a tool, a machine, a production line, an industrial plant or even various metal construction machines, works of art, etc.. It is essential to guard right from the start against this damage, hich may become catastrophic.
We must therefore be aware of the mechanism of such accidents and study their causes so that remedies can be provided.
The fight against corrosion begins with the consultancy firm and continues during assembly and finishing. It then continues during operation and successive maintenance operations whose frequency, and even the possibility of carrying them out, depend on the operating conditions: it is sometimes possible to repair and quickly replace a structural element, but it is difficult to empty a tank for its maintenance or even stop the use of a bridge or lock.
It is therefore important to able to predict this. To ensure this it is necessary to know, i.e. to have understood the process and then be able to decide what must be done before and what can be done afterwards.
DEFINITION
Corrosion can be defined as all the destructive processes which a solid body undergoes under the influence of the environment in which it is located. The actions of destruction can have several origins that are independent of each other, but are likely to occur simultaneously and therefore make an understanding of the phenomenon more difficult. This in turn makes it harder to select possible solutions. These actions may be:
- Physical
- Erosion of a pipe or valve in a liquid (water, oil) that drains into it quickly.
- If the liquid impacts on a turbine blade or a propeller, a cavitation phenomenon can occur, in addition to the erosion, causing local destruction.
- The wind (from sand, for example) may erode a protective coating, thereby attacking the part itself, such as a cable coated with zinc (several tenths of a micron).
- Chemical
A dry gas can be very corrosive, for example: oxygen (especially in the hot-rolling of steels), chlorine, fluorine or uranium hexafluoride (used in atomic power). Each product will require a specially designed coating.
- Electrochemical
This is the general case of corrosion in wet, essential object of our worries, as immersion in the atmosphere, which is never completely dry, even in temperate climates. The wetland, electrically conductive, behaves as a true electrolyte: it is capable of forming a battery between two different metals are immersed, leading to the dissolution and corrosion resistant metal only under the less noble (galvanic corrosion couple).
In these electrochemical action can be added purely chemical attack components of the solution or the environment, such as seawater, for example with particular electrolyte salt (sodium chloride)
- Biological
Some bacteria need to live with or without air (known as aerobic or anaerobic bacteria) to make interaction with the surrounding environment, and digest some elements to reject others. They can even destroy the structure of a protective coating poorly studied, and create a protected part of the attack areas simultaneously: as such, we can cite an anaerobic bacterium 'Vibrio desulfuricans "that transforms sulphate soil sulfide with oxygen and attack outside pipes buried release. Inside a water pipe, is the bacterium "Gallionela Ferruginea" causing tuber formation of hydroxides, leading to localized corrosion pitting.
In fresh water as in sea water, it quickly created on the surface of a body is immersed there a biological film on which are developing dirt that create many sources of corrosion.
On a book, in a wet air, molds (fungi) can grow, likely to reduce, then destroy the value of a paint protection while creating a particularly invisible permanent moisture.
To avoid these blooms, top coats usually containing antifouling products (tin salt, copper, weights, etc..) Is used either in shipbuilding, or "air", in a humid atmosphere ( Tropical example) on the metal art and even concrete construction works.
These different modes of destruction, physical, chemical, electrochemical, organic on all metals and non-cement and concrete, wood, fabric, plastic, etc..
The term corrosion is however the most often used for ferrous metals commonly used in construction such as steel.
MECHANISMS OF CORROSION
Surface treatments, in addition to some specific properties that motivate their choices, always provide protection against corrosion. It is therefore important to understand the behavior of a condiséré treatment against aggressive corrosive. These attacks may be biological electrochemical chemical, or erosion but electrochemical phenomena are always more or less involved.
There are microscopic heterogeneities either from metallurgical material, or the environment, or conditions of employment form a battery with one of the electrodes is more attacked than the other. The environment plays a major influence because it has a role more or less conductive electrolyte of the cell current, the electrons flowing from the most easily ionized part to another and creating a corrosion current.
When two dissimilar metals are present, battery corrosion can occur. We have indeed, in this case, two metals are electrically connected and possibly the presence of discontinuities (cracks, pits, voids) in which contact with humidity plays a more or less conductive electrolyte.
- When the coating is less "noble than the substrate, for example:
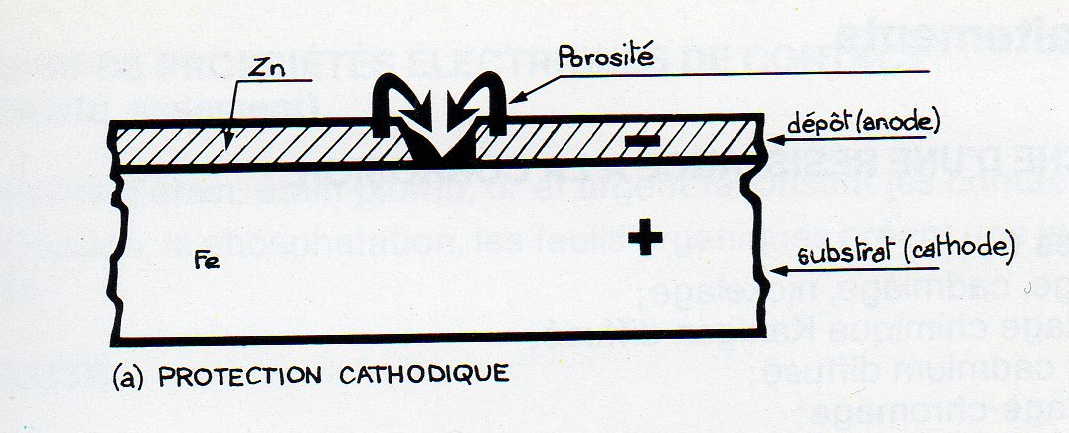 - zinc on steel (plating - plating - metal spraying);
- zinc on steel (plating - plating - metal spraying);
- Cadmium plating on steel;
- Aluminum steel metal spray gun;
said cathodic protection is: the metal base is cathode effect.
- When the coating is more "noble" than the substrate,
- Copper zinc alloy;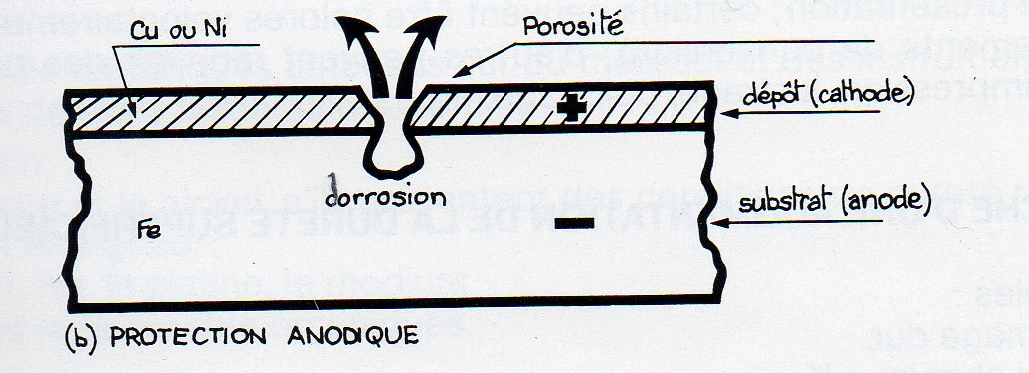
- Nickel steel;
- Electrolytic deposition of precious metals copper alloys;
Lead-steel
Anodic protection is known: the base metal is indeed anodic and cathodic coating is correlatively.
This type of corrosion is identical to that found on the bumper called "chrome" cars.
COST OF CORROSION
To evaluate this very important cost throughout the world, we must consider not only the replacement of items destroyed by corrosion, but stops functioning that cause shortfalls and sometimes significant losses examples:
- Fixed a lock gate
- Repairing a tank
- Repairing a valve pipeline
- Repairing an oil pipeline
- Repairing a manufacturing chemical fertilizer
Statistical calculations, approximate have been tempted in some countries:
- In France, corrosion represented 8-10% of the steel consumption in the 1980s, this improves.
- In the United States, at the same time, losses due to corrosion represents 2% of national income annually.
Bibliography: Raymond BENSIMON "Protection against Corrosion"







